- Home
- Details
- Registry
- RSVP
- Structure of an atom
- Centurion lounge phx
- Sherlock chapter one map
- Mapublisher scale problems
- Simon the sorcerer series
- Parallels client mac
- Motorsport manager reddit
- Abbyy ocr tool
- Bartender book
- Clan lord bestiary
- Arduino camera kit
- Junit json compare
- Multi battery pulse charger
- Pritunl gravitational
- Black friday fit bits
- Greyhound bus station
- Live doppler
- Www-ard mediathek
- Coschedule multi calendars
- Pikuniku suprematism
- Battery buddy 4098
- Madden nfl 20 ps4 gamestop
- 365 escape hospital cheat
- Michael hurricane track

Our best teachers for Grade 11 have prepared these to help you get better marks in upcoming examinations. Students of Class 11 Chemistry will be able to revise the entire chapter and also learn all important concepts based on the topic wise notes given below. Atomic Number (z) Number of protons in the nucleus of an atom.
Structure of an atom plus#
The mass number is the mass of the protons plus the mass of the neutrons.The pr esent accepted value of electrical charge is 1.6022 × 1019 C. He found that the charge on the electron to be 1.6 × 10 19 C. Millikan (1868-1953) devised a method known as oil drop experiment (1906-14), to determine the charge on the electrons. The number of protons (+) equals the number of electrons (-) in a neutral atom STRUCTURE OF ATOM 29 2.1.3 Charge on the Electron R.A.The number of protons in the nucleus is the atomic number of that atom.
Structure of an atom full#
an atom is most stable when its outermost energy level is full.
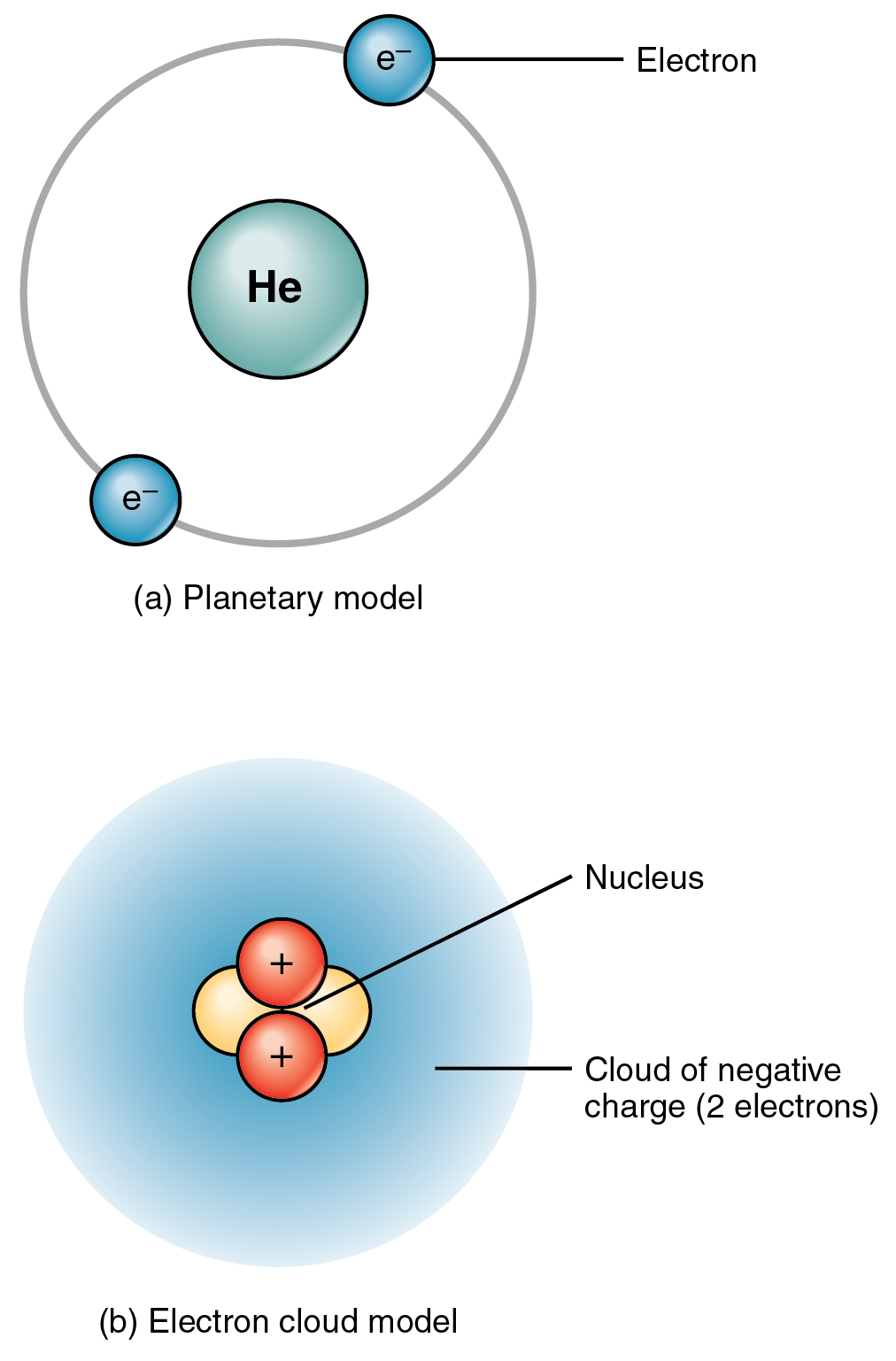
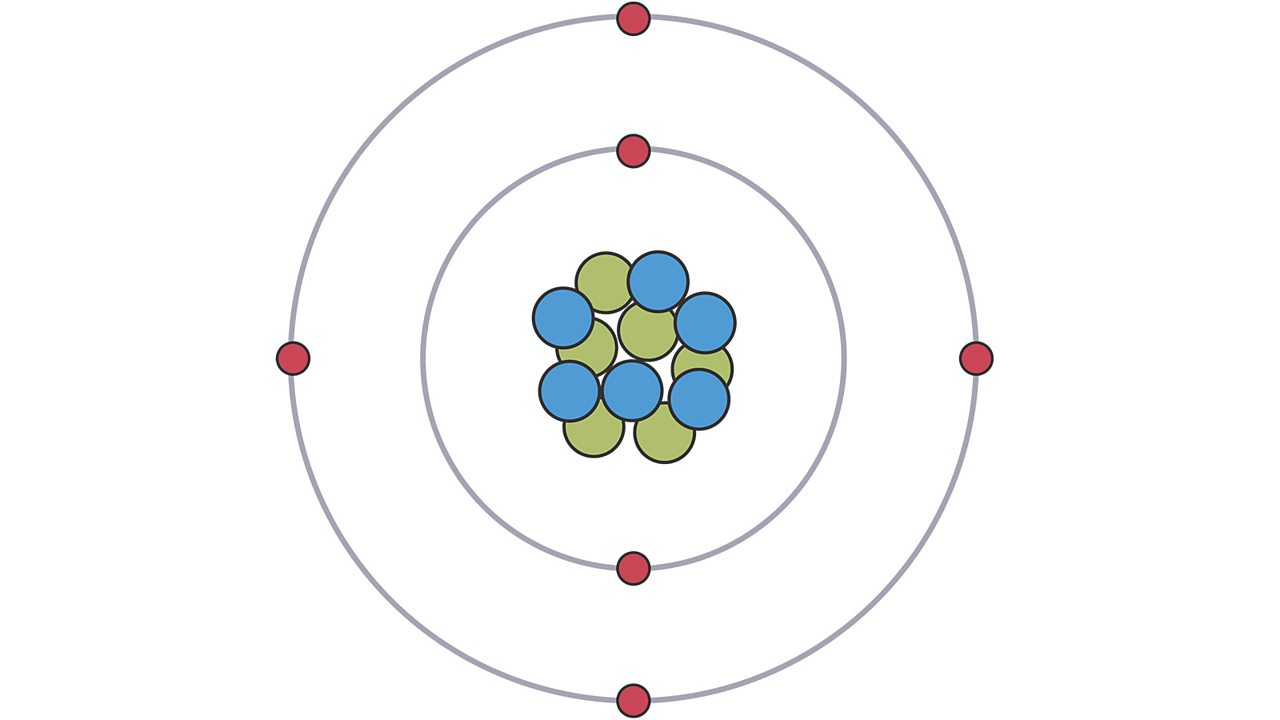
Neutrons are neutral, but protons and electrons are electrically charged. Electron cloud - an area encircling the nucleus where electrons are likely to be found Answer (1 of 6): Atoms consist of electrons surrounding a nucleus that contains protons and neutrons.The nucleus contains protons, which have a positive charge equal in magnitude to the. Neutrons (no)- particles in the nucleus that have no charge, but contribute to the atom’s mass NEUTRAL The atom consists of a tiny nucleus surrounded by moving electrons.Protons (p+)- positively charged particles in the nucleus of the atom, contribute to the atom’s mass.The nucleus contains subatomic particles that have certain characteristics:.Nucleus- Small, dense, positively charged center of the atom which contains most of the atom’s mass.Each element is composed of one type of atom and can not be broken down into a simpler substance.Atoms are composed ofsmaller subatomicparticles in an atom.Atoms have a nucleus surrounded by an electron cloud Everything in the universe is composed of individual atoms of various elements that combine together to form molecules. An atom is often referred to as the building block of matter.
- Home
- Details
- Registry
- RSVP
- Structure of an atom
- Centurion lounge phx
- Sherlock chapter one map
- Mapublisher scale problems
- Simon the sorcerer series
- Parallels client mac
- Motorsport manager reddit
- Abbyy ocr tool
- Bartender book
- Clan lord bestiary
- Arduino camera kit
- Junit json compare
- Multi battery pulse charger
- Pritunl gravitational
- Black friday fit bits
- Greyhound bus station
- Live doppler
- Www-ard mediathek
- Coschedule multi calendars
- Pikuniku suprematism
- Battery buddy 4098
- Madden nfl 20 ps4 gamestop
- 365 escape hospital cheat
- Michael hurricane track
